Technology with unknown/unlimited possibilities -living polymerization-
Introduction
Living polymerization is an effective polymerization method for synthesizing polymers with precisely controlled structures. Since the discovery of living anion polymerization in 1956, living polymerization of various mechanisms has been developed, one after another starting in the 1980s. In the 1990s, scientists discovered highly versatile living radical polymerizations, even though these reactions had previously been considered impossible with radical polymerization. Living radical polymerization has become an essential method for acquiring high-performance materials that are required in various fields, not only in the polymer industry but also in electronic and medical materials, owing to its wide range of applications.
Definition of living polymerization
Of the polymerization reactions that form polymers, chain growth polymerization generally consists of four elementary reactions, i.e., initiation, growth, stop, and chain transfer. Living polymerization is defined as a "chain growth polymerization that consists only of initiation and growth, and does not involve irreversible stop or irreversible transfer".
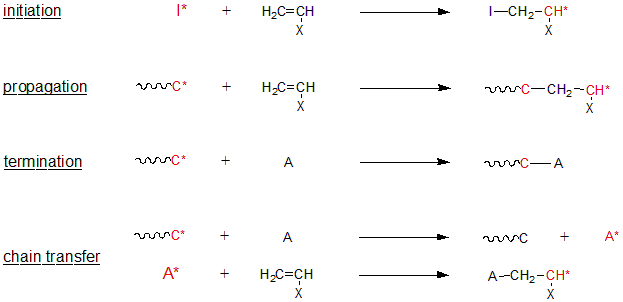
Fig. 1 Each step of chain polymerization (*: reaction active site)
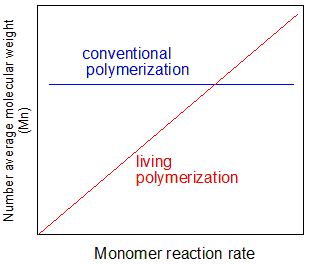
Fig. 2 Correlation between monomer reactivity and polymer molecular weight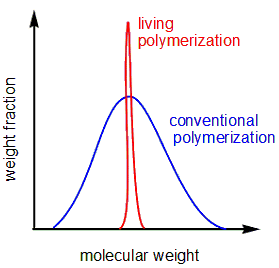
Fig. 3 Comparison of molecular weight distribution
Characteristics of living polymerization
Since the growing species continues to "live" without side reactions in living polymerization, synthesis of various polymers that could not be synthesized by the usual chain growth polymerization have been achieved. For example, it can be used to form polymers with a narrow molecular weight distribution (more uniform molecular weights), or as a simple method to form block copolymers by controlling the living terminals.
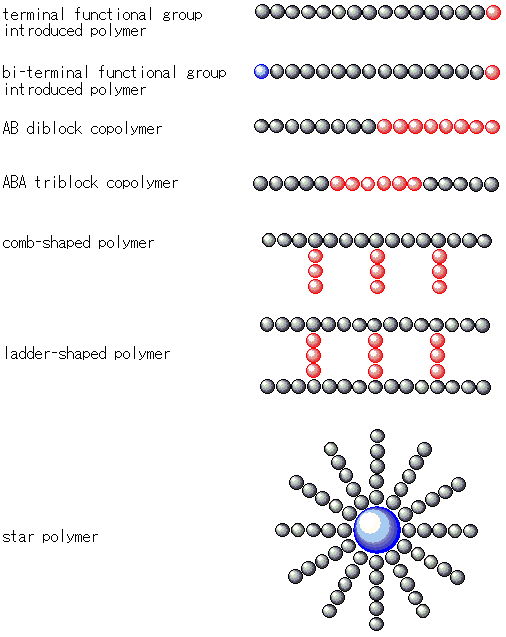
Fig. 4 Several polymer structures synthesized by living polymerization
Development of living polymerization
Living polymerization was first reported by Michael Szwarc in 1956 with anion polymerization of styrene. Szwarc introduced the concept of living polymerization in his report.
Subsequently, during the 1980s to 1990s, living polymerizations were developed as chain growth polymerizations of various mechanisms. It became possible to synthesize polymers with structures that had been difficult to form by living anion polymerization and for polymers that utilize polar monomers. In the 1990s, living polymerization was also developed for radical polymerization, which had been traditionally been considered impossible to conduct by living polymerization, owing to the presence of stable radicals, transition metal complexes, and reversible chain transfer. Today, living polymerization has been developed with various mechanisms, such as anions, cations, radicals, coordination, and ring-opening (ion, metathesis). These synthetic methods are sometimes called precision polymer synthesis, and the synthesized polymer is called a precision polymer.
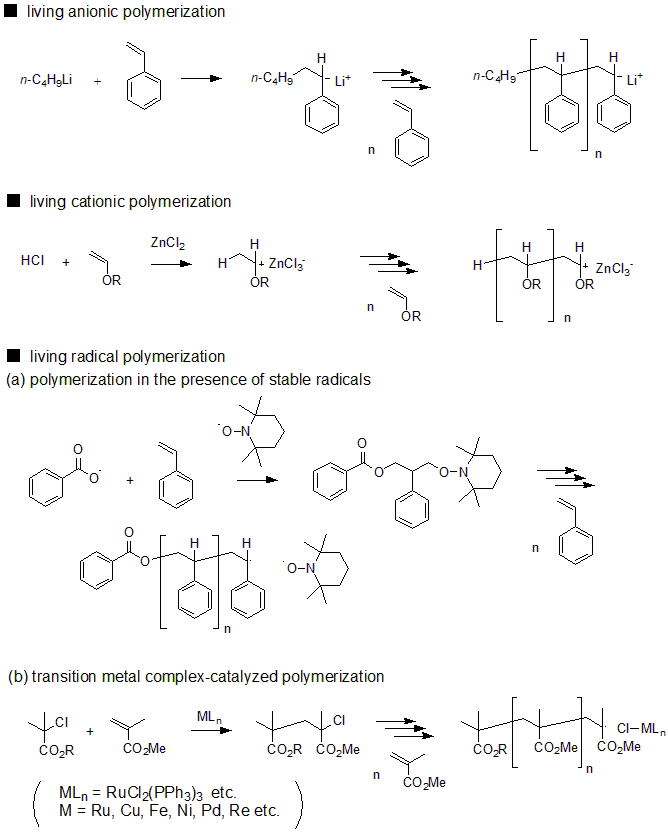
Fig. 5 Main types of living polymerization
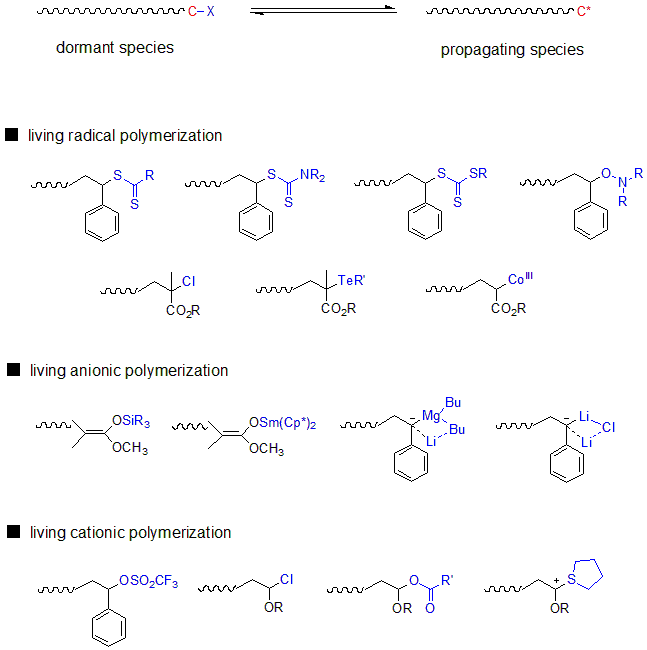
Fig.6 Dormant species (resting species) in various living polymerizations
Application of living polymerization
A body of research on the application of living polymerization, including block polymers that connect different polymer chains with covalent bonding by polymerizing different monomers from the living terminals is growing. Moreover, polymers that introduce specific functional groups at the terminals through conversion of the terminal groups are being developed into various industrial applications. Living radical polymerization in particular has been used in various applications such as photoresists, elastomers, adhesives and pressure sensitive adhesives, sealants, dispersing agents, surfactants, and self-restoration materials. More recently, applications have included biocompatible materials and biomaterials, as it does not require significant changes in the existing manufacturing facilities, has wide reproducibility and range of application, and is a polymerization method that can easily be applied to various functional monomers.




