Anti α-Synuclein Antibodies
Diseases characterized by the accumulation of α-synuclein are called "synucleinopathies," and examples include Parkinson's disease, dementia with Lewy bodies, and multiple system atrophy. In 1997, mutations in the α-synuclein gene were identified as the cause of familial Parkinson's disease. Subsequently, α-synuclein was shown to be the principal component of Lewy bodies found in the brains of patients with sporadic Parkinson's disease. These findings focused significant attention on the relationship between Parkinson's disease and α-synuclein. Notably, nearly all accumulated α-synuclein is phosphorylated, and ongoing research is investigating how the phosphorylation of α-synuclein contributes to the onset and progression of Parkinson's disease.
Fujifilm Wako offers mouse monoclonal antibodies that specifically react with Ser129-phosphorylated α-synuclein. These product do not react with unphosphorylated α-synuclein and selectively recognize only accumulated Ser129-phosphorylated α-synuclein. Accordingly, it is suitable for visualizing Lewy bodies by immunohistochemistry and for detecting Ser129-phosphorylated α-synuclein by Western blotting.
What is α-Synuclein?
α-Synuclein (α-Syn) is a 140-amino acid protein. In humans, three synuclein isoforms have been identified: α-, β-, and γ-synuclein. Both α- and β-synuclein are abundantly expressed in the presynaptic terminals in the central nervous system1). In contrast, γ-synuclein is scarcely expressed in the nervous system and its association with breast and ovarian cancers has been reported2-3). Although the precise physiological functions of synucleins have not yet been fully elucidated, α-synuclein is thought to be involved in the regulation of neurotransmitter release4) and the formation and clustering of synaptic vesicles5-6).
α-Synuclein readily undergoes fibrillization and is believed to accumulate in the brain in its aggregated form, where it exerts cytotoxic effects. Disorders characterized by the accumulation of α-synuclein are collectively referred to as synucleinopathies, including Parkinson’s disease, dementia with Lewy bodies, and multiple system atrophy. In 1997, mutations in the α-synuclein gene were identified as the cause of familial Parkinson’s disease7). Subsequently, α-synuclein was shown to be the principal component of Lewy bodies found in the brains of patients with sporadic Parkinson’s disease8-9). These findings focused significant attention on the relationship between Parkinson’s disease and α-synuclein. Biochemical analyses have further revealed that α-synuclein present in Lewy bodies undergoes post-translational modifications such as phosphorylation and ubiquitination. Notably, nearly all accumulated α-synuclein is phosphorylated, and ongoing research is investigating how the phosphorylation of α-synuclein contributes to the onset and progression of Parkinson’s disease.
Previous studies suggest that fibrillized and abnormally accumulated α-synuclein may trigger neuronal cell death, which may be one of the underlying causes of Parkinson’s disease. However, it remains unclear whether the primary cytotoxic species is aggregated α-synuclein itself, intermediate species formed during aggregation, or earlier-stage α-synuclein fibrils. In recent years, α-synuclein fibrils have been reported to propagate between cells10), with extracellular vesicles (EVs), such as exosomes, potentially playing a role in this transmission11). Such intercellular spread is thought to be a critical step in the onset and progression of Parkinson’s disease, and further elucidation of its underlying mechanisms is highly anticipated.
Anti Phosphorylated α-Synuclein, Monoclonal Antibody (pSyn#64)
This product is a mouse monoclonal antibody that specifically reacts with Ser129-phosphorylated α-synuclein. α-Synuclein found in Lewy bodies of patients with Parkinson’s disease and dementia with Lewy bodies is known to be phosphorylated at Ser12912-13). This product does not react with unphosphorylated α-synuclein and selectively recognizes only accumulated Ser129-phosphorylated α-synuclein. Accordingly, it is suitable for visualizing Lewy bodies by immunohistochemistry and for detecting Ser129-phosphorylated α-synuclein by Western blotting.
Antibody Information
| Clonality | Monoclonal (Clone No. pSyn#64) |
|---|---|
| Antigen | Human α-synuclein (residues 124–134) phosphorylated at Ser129 |
| Host | Mouse |
| Formulation | Cell culture supernatant containing serum, 0.05% sodium azide |
| Conjugate | Unconjugated |
| Cross-reactivity | Human, Mouse, Rat (α-synuclein phosphorylated at Ser129) |
| Application | Immunohistochemistry 1:1,000-10,000 Western blotting 1:1,000-10,000 |
Application Data
Immunohistochemistry
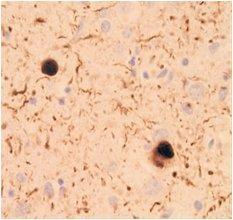
Species: Human (Dementia with Lewy bodies)
Site: Brain
Data by courtesy of
Dr. Iwatsubo, Graduate School of Medicine, The University of Tokyo
Detection of Ser129-phosphorylated α-synuclein in skin biopsy specimens from patients with synucleinopathies
Gibbons et al. (JAMA, 331(15), 1298 (2024))14) investigated whether synucleinopathies—such as Parkinson’s disease, multiple system atrophy, and dementia with Lewy bodies—could be detected by analyzing α-synuclein in skin biopsy specimens using Fujifilm Wako’s Anti Phosphorylated α-Synuclein, Monoclonal Antibody (pSyn#64).
| Diagnosis | P-SYN positive*1 | P-SYN negative*2 | Proportion of participants positive for P-SYN (95% CI) |
|
|---|---|---|---|---|
| Synucleinopathies | 213 | 10 | 95.5% (91.9 - 97.8%) | |
| Parkinson’s disease | 89 | 7 | 92.7% (85.6 - 97.0%) | |
| Multiple system atrophy | 54 | 1 | 98.2% (91.7 - 99.9%) | |
| Dementia with Lewy bodies | 48 | 2 | 96.0% (86.3 - 99.5%) | |
| Pure autonomic failure | 22 | 0 | 100% (84.6 - 100%) | |
| No synucleinopathies | 4 | 116 | 3.3% (1.3 - 8.0%) | |
(Based on Reference 14)
*1 Positive for P-SYN in at least one biopsy sites.
*2 Negative for P-SYN in all biopsy sites.
[Result]
The proportion of skin biopsy specimens positive for Ser129-phosphorylated α-synuclein was 92.7% in Parkinson’s disease, 98.2% in multiple system atrophy, 96.0% in dementia with Lewy bodies, and 100% in pure autonomic failure, whereas only 3.3% of individuals without a history of synucleinopathy (control group) tested positive (Table 1).
Anti Phosphorylated α-Synuclein, Monoclonal Antibody(pSyn#64), Biotin-conjugated
This product is a biotin-conjugated anti-Ser129-phosphorylated α-synuclein monoclonal antibody (pSyn#64). It enables sensitive detection of Ser129-phosphorylated α-synuclein with low background using techniques such as the avidin-biotin complex (ABC) method in combination with peroxidase-conjugated streptavidin.
Antibody Information
| Clonality | Monoclonal (Clone No. pSyn#64) |
|---|---|
| Antigen | Human α-synuclein (residues 124–134) phosphorylated at Ser129 |
| Host | Mouse |
| Formulation | D-PBS, 0.05% sodium azide |
| Conjugate | Biotin-conjugated |
| Cross-reactivity | Human, Mouse, Rat (α-synuclein phosphorylated at Ser129) |
| Application | Immunocytochemistry 1:200-5,000 Immunohistochemistry 1:200-5,000 |
Application Data
Immunohistochemistry
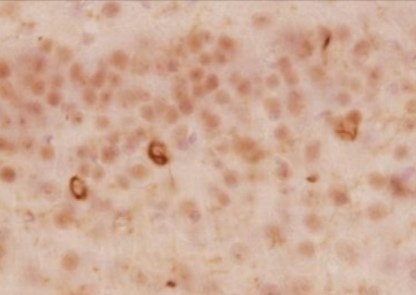
+
Biotin-conjugated secondary antibody
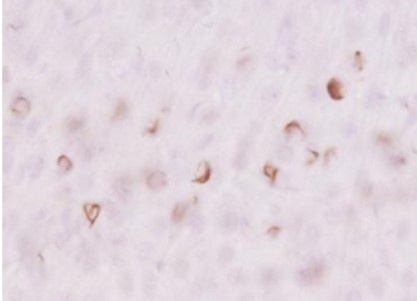
Species: Mouse (6 months old, wild-type, injected intracerebrally with aggregated α-synuclein)
Site: Striate
Sample: Vibratome section (50 μm thick)
Antibody concentration: 1:1,000
Staining: DAB staining
Data by courtesy of
Drs. Kuwahara and Iwatsubo, Graduate School of Medicine, The University of Tokyo
[Result]
By combining the biotin-labeled antibody with the ABC method, background staining was reduced compared with detection using a secondary antibody, resulting in clearer, more distinct staining.
Bulk Supply of Antibody
Fujifilm Wako also offers bulk supply of this product (mg scale). Testing for antibody activity (titer) and protein concentration can also be performed upon request. For inquiries, please contact us at .
- Please note that, depending on your specific requirements, bulk supply may not be available, or delivery may take additional time.
- Certain products may not be available for commercial use. For commercial applications, please contact Fujifilm Wako prior to such use.
参考文献
- Goedert, M.: Nat. Rev. Neurosci., 2(7), 492(2001).
Alpha-synuclein and neurodegenerative diseases - Ji, H. et al.: Cancer Res., 57(4), 759(1997).
Identification of a breast cancer-specific gene, BCSG1, by direct differential cDNA sequencing - Jia, T. et al.: Cancer Res., 59(3), 742(1999).
Stimulation of breast cancer invasion and metastasis by synuclein γ - Chandra, S. et al.: Proc. Natl. Acad. Sci. USA, 101(41), 14966(2004).
Double-knockout mice for alpha- and beta-synucleins: effect on synaptic functions - Burré, J. et al.: Science, 329(5999), 1663(2010).
Alpha-synuclein promotes SNARE-complex assembly in vivo and in vitro - Suzuki, C. et al.: Biochim. Biophys. Acta Mol. Basis Dis., 1870(8), 167494(2024).
Direct evidence for ultrastructures of the α-synuclein-associated synaptic vesicle pool in presynaptic terminals - Polymeropoulos, M. H. et al.: Science, 276(5321), 2045(1997).
Mutation in the alpha-synuclein gene identified in families with Parkinson's disease - Baba, M. et al.: Am. J. Pathol., 152(4), 879(1998).
Aggregation of alpha-synuclein in Lewy bodies of sporadic Parkinson's disease and dementia with Lewy bodies - Spillantini, M. G. et al.: Nature, 388(6645), (1997).
Alpha-synuclein in Lewy bodies - Li, J. Y. et al.: Nat. Med., 14(5), 501(2008).
Lewy bodies in grafted neurons in subjects with Parkinson's disease suggest host-to-graft disease propagation - Stuendl, A. et al.: Brain, 139, 481(2016).
Induction of α-synuclein aggregate formation by CSF exosomes from patients with Parkinson's disease and dementia with Lewy bodies - Fujiwara, H. et al.: Nat. Cell Biol., 4(2), 160(2002).
alpha-Synuclein is phosphorylated in synucleinopathy lesions - Saito, Y. et al.: J. Neuropathol. Exp. Neurol., 62(6), 644(2003).
Accumulation of phosphorylated alpha-synuclein in aging human brain - Gibbons, C. H. et al.: JAMA, 331(15), 1298(2024).
Skin Biopsy Detection of Phosphorylated α-Synuclein in Patients With Synucleinopathies
Product List
- Open All
- Close All
Anti Phosphorylated α-Synuclein, Monoclonal Antibody (pSyn#64)
Anti Phosphorylated α-Synuclein, Monoclonal Antibody(pSyn#64), Biotin-conjugated
For research use or further manufacturing use only. Not for use in diagnostic procedures.
Product content may differ from the actual image due to minor specification changes etc.
If the revision of product standards and packaging standards has been made, there is a case where the actual product specifications and images are different.



