Deuterated Solvents
Selection of Deuterated Solvent for qNMR
The quality of the deuterated solvent is one of the extremely important components in qNMR applications, as is the case with the calibration standard. If the impurity signal contained in the deuterated solvent overlaps with the target signal, it will be a bias in the signal area for the qNMR measurement. In addition, when a huge signal generated from water or non-deuterated functional group (e.g., non-deuterated methyl group of acetone-d6) in the deuterated solvent is observed, optimal sensitivity settings (adjustment of receiver gain) in NMR measurement affects the dynamic range, and as a result, the sensitivity of the target signals (i.e., the signals of analyte and calibrat) becomes lowered.
What are the cautionary points when selecting the deuterated solvent used for qNMR?
- Impurity signal: impurity signal overlaps with the signal to be used for quantification, which lowers the accuracy of qNMR results.
- Moisture content: signals of water may affect the dynamic range and lower the sensitivity of the signal you want to use for quantification.
- Deuteration ratio: deuterated signal may affect the dynamic range and lower the sensitivity of the signal you want to use for quantification.
So far, users mainly tended to judge the quality of deuterated solvents by the deuteration ratio. However, due to the recent improvement in the sensitivity of NMR instrumentation and NMR data acquisition under quantitative conditions due to the dissemination of the qNMR technique. There is increasing concern about impurities generated from deuterated solvents. As with HPLC and GC analysis, the supply of deuterated solvents with high-purity and quality is beginning to be required for qNMR measurement.
In consideration of the above points, FUJIFILM Wako developed high-purity NMR solvents suitable for qNMR measurement. For the high-purity NMR solvent, we guaranteed the quality by limit test for impurities with 1H, 13C, 19F and 31P NMR spectra acquired under high-sensitivity data acquisition parameters in addition to chemical purity with GC-FID (except deuterium oxide). In addition, we have set acceptance criteria such as low moisture content and high deuteration ratio.
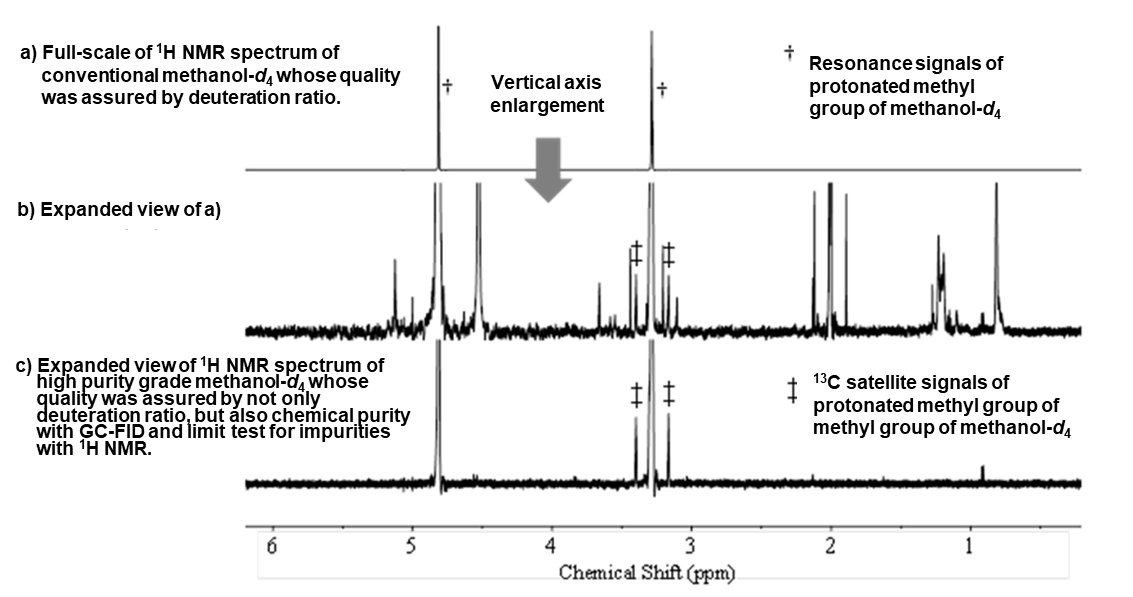
High Purity NMR Solvent
This series is a solvent for NMR measurement with high purity that reduces impurities that affect NMR measurement. No signal other than water and protium solvent* is observed in those products. This products do not contain impurities of other deuterium solvents and guarantees a low water content. So it is useful when conducting quality tests of pharmaceutical products and quantitative NMR (qNMR).
*A signal with an intensity difference of 30% or more from the baseline.
Specification [Chloroform-d, 99.8% (High purity)]
| Requirement | Specification |
|---|---|
| Appearance | Colorless clear liquid |
| Water | max 0.003% |
| Degree of deuteration | min. 99.8% |
| Assay (capillary GC) | min. 99.9% |
| Impurities (NMR) (1H) | to pass test |
| Impurities (NMR) (13C) | to pass test |
| Impurities(NMR) (31P) | to pass test |
| Impurities (NMR) (19F) | to pass test |
1H NMR chart (Chloroform-d)
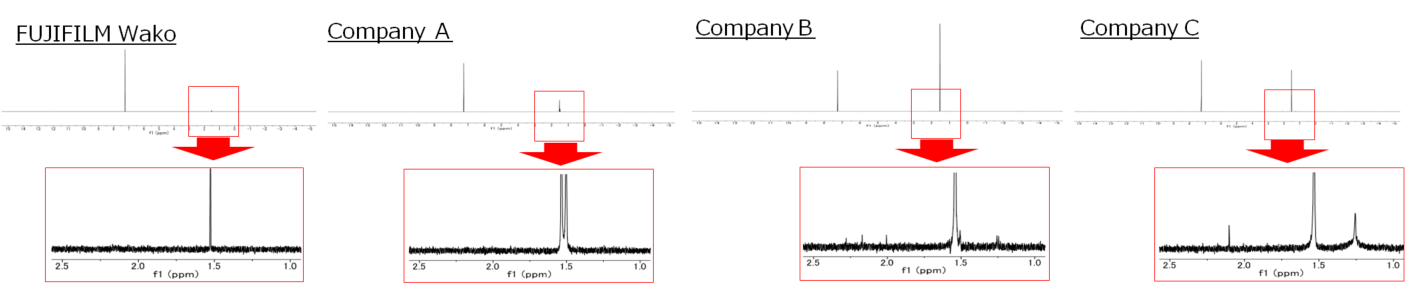
| Chloroform-d | ||||
|---|---|---|---|---|
| FUJIFILM Wako | Company A | Company B | Company C | |
| Water | max. 0.003% | 0.005 % (CoA) max. 0.02% |
0.0013 % (CoA) max. 0.01% |
0.0015 % (CoA) max. 0.01% |
| Assay | min. 99.9% | 100 % (CoA) | min. 99% | min. 99% |
| Impurities (NMR) (1H) | to pass test | Water peak (1.49 ppm) |
Impurity peaks (1.24, 1.51, 2.00, 2.16 ppm) |
Impurity peaks (1.25, 1.51, 2.10ppm) |
| Impurities (NMR) (13C) | to pass test | - | - | - |
| Impurities (NMR) (31P) | to pass test | |||
| Impurities (NMR) (19F) | to pass test | |||
Product List
- Open All
- Close All
High Purity NMR Solvents
General Grade
For research use or further manufacturing use only. Not for use in diagnostic procedures.
Product content may differ from the actual image due to minor specification changes etc.
If the revision of product standards and packaging standards has been made, there is a case where the actual product specifications and images are different.




