Fludioxonil, Azoxystrobin, and Pyrimethanil
Fludioxonil, azoxystrobin, and pyrimethanil are designated as food additives (fungicides) by the "Ordinance for Enforcement of the Food Sanitation Act" and "Specifications and Standards for Food, Food Additives, etc. (Ministry of Health and Welfare Notification No. 370, 1959)."
Specifications and standards for fludioxonil, azoxystrobin, and pyrimethanil as food additives have been established. Quantitative NMR (qNMR)*1 has been used for analyzing the purity of the reference materials for assay.
FUJIFILM Wako offers reference materials*2 and analytical standard whose purity has been evaluated by qNMR*3.
- 1: In this evaluation method, purity or concentration of the sample is evaluated using a reference material assigned with a value as the standard of the amount of the substance.
- 2: FUJIFILM Wako has accredited as a reference material producer of National Institute of Technology and Evaluation International Accreditation Japan (NITE IA JAPAN); a certificate with the "ASNITE" logo is attached to the products.
- 3: Pyrimethanil is the analytical standard (for pesticide residue analysis) guaranteed by FUJIFILM Wako.
Overview
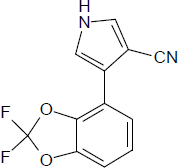
Fludioxonil Reference Material
|
CAS RN®: 131341-86-1 C12H6F2N2O2 = 248.19 |
 |
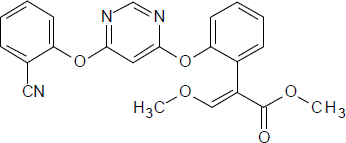
Azoxystrobin Reference Material
|
CAS RN®: 131860-33-8 C22H17N3O5 = 403.39 |
 |
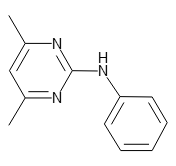
Pyrimethanil Standard
|
CAS RN®: 53112-28-0 C12H13N3 = 199.25 |
 |
Product List
- Open All
- Close All
For research use or further manufacturing use only. Not for use in diagnostic procedures.
Product content may differ from the actual image due to minor specification changes etc.
If the revision of product standards and packaging standards has been made, there is a case where the actual product specifications and images are different.



