Water-soluble Acrylamide Monomers
Polymers made from acrylamide monomers are used in various applications, including UV paints, adhesives, and lens materials. Acrylamide polymers have recently received much attention as biomaterials, including cell culture sheets, due to their high biocompatibility. Here we introduce an acrylamide monomer and cross-linkers with multiple acrylamide groups, that are both water-soluble and safe.
Features
- It has excellent water- solubility and safety.
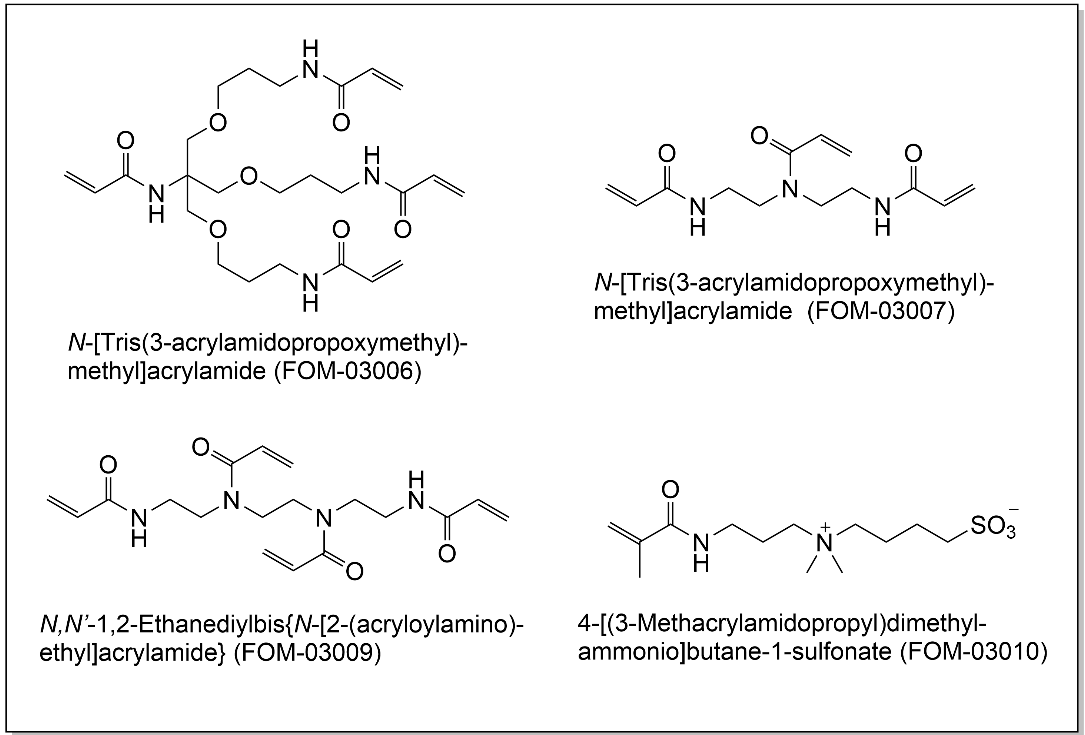
- Four cross-linkers with 2- to 4 acrylamide groups per molecule (FOM-03006 ~ 03009).
- One monomer with excellent hydrophilicity derived from a betaine structure (FOM-03010).
- Try to be used for research onto study biocompatible materials, paints, adhesives, inks, cleaners, lens materials, flocculants, etc.
- Support for mass production is possible.
About polyacryamide
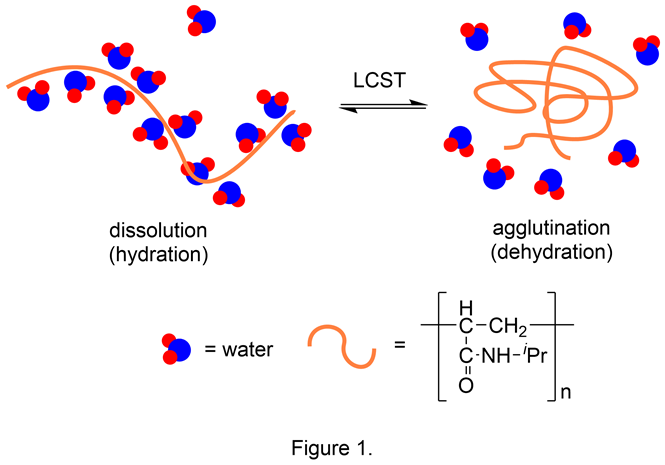
Acrylamide polymers dissolve in water through hydration by hydrogen bonding between the amide carbonyl groups and water molecules. While they are widely used in UV paints, adhesives, lens materials, superabsorbents, flocculants, and oil recovery agents, they have drawn attention as biomaterials due to their high biocompatibility. Poly(N-isopropylacrylamide) (PIPAAm) is a polymer with a lower critical solution temperature (LCST) of 32℃ in water. It forms a contracted structure due to dehydration at 32℃ or higher and swells to dissolve in water due to hydration at 32℃ or lower (Figure 1). Using these excellent features, PIPAAm has been applied to cell culturewares used to prepare cell sheets.1) In order to make these cell culturewares, PIPAAm is grafted in nanometer orders on a polystyrene substrate. The PIPAAm layer allows adhesion of cells due to its hydrophobicity, at 37℃ (the same temperature used for cell culture); however, as it cools to room temperature, it becomes hydrophilic and shows noninvasive recovery of cells cultured on the surface. Further, these cell culturewares are also used to culture a wide variety of cell species such as corneal epithelial cells, oral mucosal epithelial cells, fibroblasts, and myoblasts.
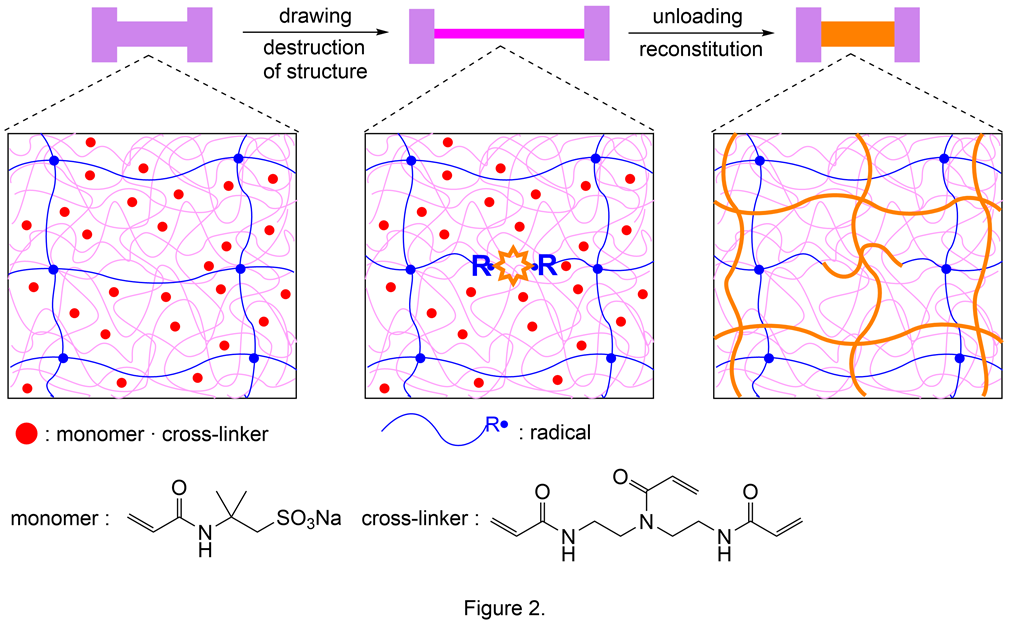
A polymer material characterized by its mechanical strength increased by applying mechanical load like human muscles has been developed using polyacrylamide.2), 3) Human muscles can grow and increase their strength by restructuring them through repeated construction/destruction processes by incorporating nutrients from outside. In order to develop artificial materials like human muscles, Gong and Nakajima et al. focused on double network gels (DN gels) capable of incorporating substances from outside the solvent and producing radicals upon the destruction of fragile polymer networks. The mechanism of polymer restructuring using DN gel is described as follows:
- The mechanical load applied to DN gel containing monomers and cross-linkers destroys its fragile macromolecular net (breakdown of the structure).
- At the same time, radicals are generated and radical polymerization of the internal monomers and cross-linkers proceeds to synthesize a new macromolecular net (reformation of the structure), thereby increasing the strength of the gel (Figure 2).
In fact, radical polymerization in DN gel associated with radical formation was confirmed when DN gel was immersed into a solution of monomer (2-acrylamide-2-methylpropanesulfonic acid) and cross-linker (N,N-bis(2-acrylamideethyl)acrylamide) and then stretched in an argon atmosphere. The monomer conversion rate in this reaction was 80% or higher, and stretching increased the polymer mass in DN gel by approximately three times. Repeated stretching of DN gel in the monomer/cross-linker solution has also been confirmed to increase its weight, strength, and hardness. This material is the world's first example of an “organism-like material” that incorporates nutrients from outside and grows.
References
- Hashimoto, S:Polymers, 66, 499 (2017).
- Nakajima, Y : Polymers, 69, 146 (2020).
- Matsuda, T., Kawakami, R., Namba, R., Nakajima, T. and Gong, J. P. : Science., 363, 504 (2019).
- Gong, J. P., Katsuyama, Y., Kurosawa, T., Osada, Y. : Adv. Mater., 15, 1155 (2003).
Physical characteristic and feature of water-soluble cross-linkers
N-[Tris(3-acrylamidopropoxymethyl)methyl]acrylamide (another name: FOM-03006)
Structure
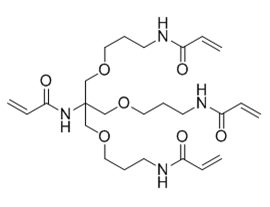
Physical Characteristic
| Appearance | White solid |
|---|---|
| Melting point | 107℃ |
| Soluble solvent | Methanol |
| Water-soluble | Soluble |
Features
1. Highly water-soluble
It exhibits high solubility in water and readily soluble in hydrophilic solvents such as methanol and ethanol.
Solubility in various solvent
| Solvent | SP | Solubility at monomer concentration (wt%) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| 0.1 | 0.5 | 1 | 10 | 20 | 30 | 40 | 50 | ||
| n-Hexane | 7.3 | × | ー | ー | ー | ー | ー | ー | ー |
| Toluene | 8.8 | × | ー | ー | ー | ー | ー | ー | ー |
| Ethyl acetate | 9.0 | × | ー | ー | ー | ー | ー | ー | ー |
| 2-Butanone | 9.3 | 〇 | × | ー | ー | ー | ー | ー | ー |
| Acetone | 10.0 | 〇 | 〇 | × | ー | ー | ー | ー | ー |
| 2-Propanol | 11.5 | ◎ | ◎ | ◎ | ◎ | 〇 | 〇 | 〇 | △ |
| Acetonitrile | 11.9 | 〇 | 〇 | × | ー | ー | ー | ー | ー |
| Ethanol | 12.7 | ◎ | ◎ | ◎ | ◎ | ◎ | 〇 | 〇 | 〇 |
| Methanol | 14.5 | ◎ | ◎ | ◎ | ◎ | ◎ | ◎ | 〇 | 〇 |
| Water | 23.4 | ◎ | ◎ | ◎ | ◎ | 〇 | 〇 | 〇 | 〇 |
*SP: Solubility Parameter, SP of monomer = 12.8
*Solubility is measured at 30℃.
*◎: Easy 〇: Food △: Solid precipitation over time ×: No soluble -: No measurement
2. Excellent resistance to hydrolysis
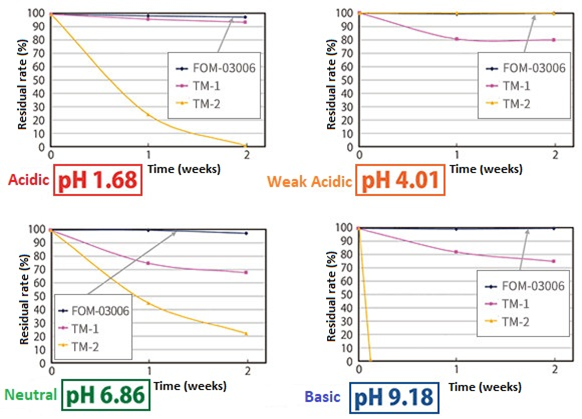
It has excellent resistance to hydrolysis in acidic to basic aqueous solutions.
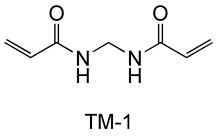
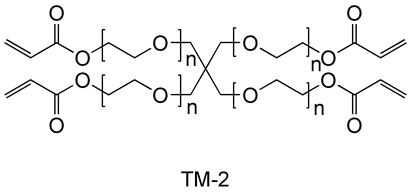
The stability of N-[tris(3-acrylamidepropoxymethyl)methyl]acrylamide (FOM-03006) in an aqueous solution was evaluated (45℃, 2 weeks). Commercially available bifunctional acrylamide TM-1 and EO denatured tetrafunctional acrylate TM-2 are used as comparators.
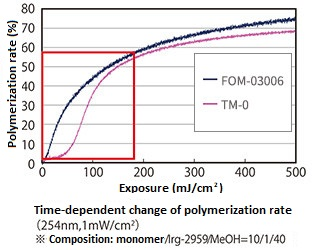
3. High curability
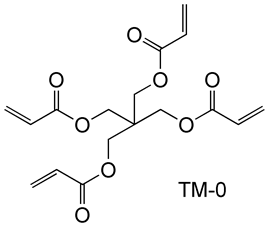
It also has high curability and less susceptible to inhibit polymerization by oxygen, and exhibits a higher degree of polymerization than commercially available tetrafunctional acrylate TM-0.
4. Excellent safty
It has an excellent level of safety, unlike conventional acrylamide.
Safty test of N-[tris(3-acrylamidepropoxymethyl)methyl]acrylamide
| Test items | Results |
|---|---|
| Skin irritation and corrosiveness | PII = 0 |
| non-stimulating | |
| Skin sensitization | negative |
| Mutagenicity (Ames) | negative |
Examples of curing
Curing example 1: Example of radical polymerization
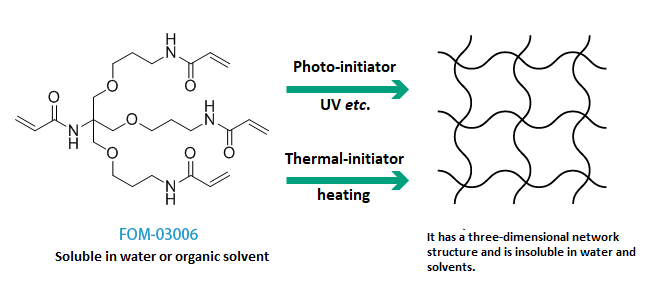
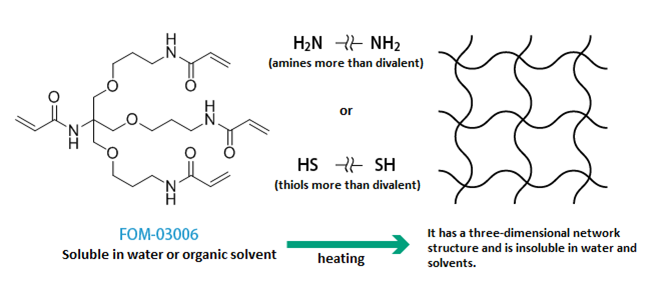
Curing example 2: Example of Michael addition reaction
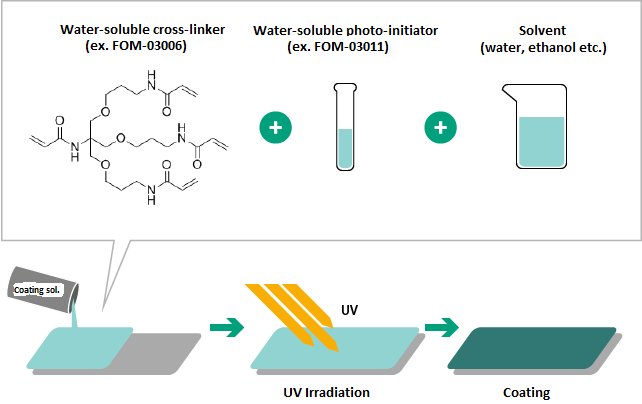
Example of use
Example 1
- Highly hydrophilic functional coatings were prepared.
- Due to the high water-solubility of monomers and initiators, water may be used as a solvent to prepare coating solutions.
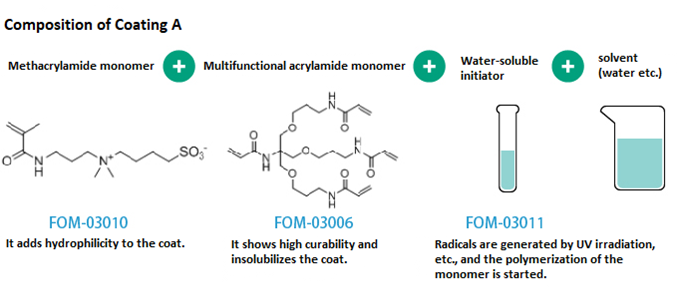
Example composition of Coating A
| Materials | Mixing ratio (wt%) |
|---|---|
| Water-soluble monomer (FOM-03010) | 30 |
| Water-soluble cross-linker (FOM-03006) | 67 |
| Water-soluble photoradical initiator (FOM-03011) | 3 |
| Methanol | 400 |
Examples of use and physical properties of Coating A
- After coating to a PET substrate and being dried (50℃/5 min), photocured (3 J/cm 2).
*Lamp: High-pressure mercury lamp (Exposure is controlled at UV-A)
| Film | Water contact angle (°) |
|---|---|
| PET only | 75 |
| Coating A | 35 |
| Soak the Coating A with water and dry | 44 |
In evaluation of wettability using water contact angle as an indicator of affinity with water, Coating A has a low contact angle indicating high hydrophilicity and maintains its hydrophilicity even when re-dried after immersion in water.
Example 2.
- A coating film less susceptible to curling with a lower curing shrinkage rate can be prepared to replace a commercially available polyfunctional acrylic monomer with FOM-03006.
Composition of coating solution
| Coating B | Coating C | Mixing ratio (wt%) |
|---|---|---|
| Water-soluble cross-linker (FOM-03006) | Commercially available polyfunctional monomer | 50 |
| Monofunctional methacryl monomer (HEMA) | Monofunctional methacryl monomer (HEMA) | 47 |
| Water-soluble photoradical initiator (FOM-03011) | Water-soluble photoradical initiator (FOM-03011) | 3 |
State of coating films after curing

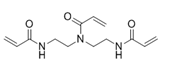
N,N-Bis(2-acrylamidoethyl)acrylamide (another name: FOM-03007)
Structure
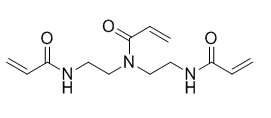
Physical characteristic
| Appearance | White solid |
|---|---|
| Melting point | 89℃ |
| Soluble solvent | Methanol |
| Water-soluble | Soluble |
Solubility in various solvent and monomer
| Solvent / Monomer | Solubility (wt%) |
|---|---|
| n-Hexane | <0.1 |
| Toluene | <0.1 |
| Ethyl acetate | 0.1 ~ 0.5 |
| 2-Butanone | 0.1 ~ 0.5 |
| Acetone | 0.1 ~ 0.5 |
| 2-Propanol | 0.5 ~ 1 |
| Acetonitrile | <0.1 |
| Ethanol | ≧50 |
| Methanol | ≧50 |
| 1-Methoxy-2-propanol | 10 ~ 20 |
| Water | ≧50 |
| 2-Hydroxyethyl methacrylate (HEMA) | 30 ~ 40 |
| N-(2-Hydroxyethyl)acrylamide | 30 ~ 40 |
| N,N-Dimethylacrylamide | 20 ~ 30 |
| 4-Acryloylmorpholine | 5 ~ 10 |
*Measured at 30℃
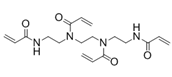
N,N'-1,2-Ethanediylbis{N-[2-(acryloylamino)ethyl]acrylamide} (another name: FOM-03009)
Structure
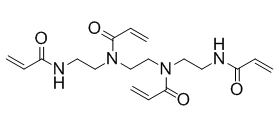
Physical characteristic
| Appearance | White powder |
|---|---|
| Melting point | 89℃ |
| Soluble solvent | Methanol |
| Water-soluble | Soluble |
Solubility in various solvent and monomer
| Solvent / Monomer | Solubility (wt%) |
|---|---|
| n-Hexane | <0.1 |
| Toluene | <0.1 |
| Ethyl acetate | 0.1 ~ 0.5 |
| 2-Butanone | 1 ~ 5 |
| Acetone | 1 ~ 5 |
| 2-Propanol | 1 ~ 5 |
| Acetonitrile | 1 ~ 5 |
| Ethanol | 30 ~ 40 |
| Methanol | ≧50 |
| 1-Methoxy-2-propanol | 10 ~ 20 |
| Water | 40 ~ 50 |
| 2-Hydroxyethyl methacrylate (HEMA) | 10 ~ 20 |
| N-(2-Hydroxyethyl)acrylamide | 20 ~ 30 |
| N,N-Dimethylacrylamide | 5 ~ 10 |
| 4-Acryloylmorpholine | 5 ~ 10 |
*Measured at 30℃
Summary of physical characteristic about cross-linkers
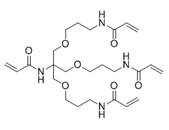 |
 |
 |
|
| Appearance | White solid | White solid | White powder |
|---|---|---|---|
| Melting point | 107℃ | 89℃ | 110℃ |
| Soluble solvent | Water, Methanol, Ethanol | Water, Methanol, Ethanol | Water,Methanol |
Physical characteristic and feature of water-soluble monomer
4-[(3-Methacrylamidopropyl)dimethylammonio]butane-1-sulfonate (another name: FOM-03010)
FOM-03010 is water-solble monomer, which has a highly hydrophilic betaine structure with methacrylamide groups. It has high water-solubility, biocompatible, and safety for human body.
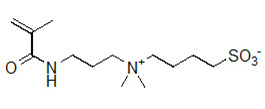
Physical characteristic
| Appearance | White powder |
|---|---|
| Melting point | - |
| Odor | Odorless |
Features
1. High water-solubility and hydrophilicity
- Monomer is soluble in water with a solubility of at least 50 wt%. Moreover, it is readily soluble in solvents such as methanol and ethanol.
- Highly hydrophilic due to the presence of a betaine structure (CLogP = -10).
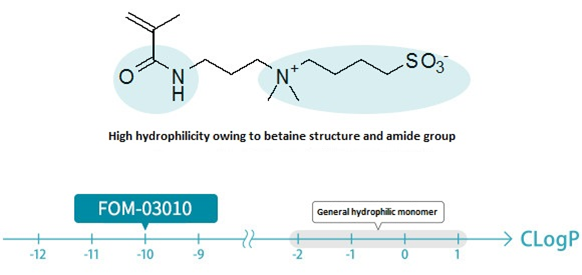
Many of the hydrophilic monomers presently available in the market have CLogP values of around −2 to + 1, a parameter used as an indicator of hydrophilicity/hydrophobicity. FOM-03010 has a CLogP value of −10 and is much more hydrophilic than these general-purpose monomers.
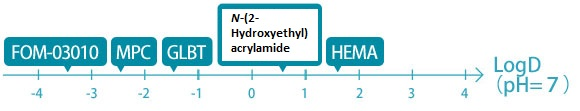
GLBT: Carboxymethylbetaine monomer
HEMA: 2-Hydroxyethyl methacrylate
FOM-03010 shows high hydrophilicity in terms of LogD = −4 (pH = 7), an indicator of hydrophilicity defined by considering the dissociation of molecules under different pH environments.
Solubility in various solvent and monomer
| Solvent・Monomer | Solubility at monomer concentration (wt%) | |||||||
|---|---|---|---|---|---|---|---|---|
| 0.1 | 0.5 | 1 | 10 | 20 | 30 | 40 | 50 | |
| Water | 〇 | 〇 | 〇 | 〇 | 〇 | 〇 | 〇 | 〇 |
| Methanol | 〇 | 〇 | 〇 | 〇 | 〇 | 〇 | × | - |
| Ethanol | 〇 | 〇 | 〇 | 〇 | × | - | - | - |
| 2-Propanol | 〇 | 〇 | × | - | - | - | - | - |
| Dimethyl sulfoxide | 〇 | 〇 | 〇 | 〇 | 〇 | × | - | - |
| Acetone | × | - | - | - | - | - | - | - |
| Ethyl acetate | × | - | - | - | - | - | - | - |
| N,N-Dimethylacetamide | × | - | - | - | - | - | - | - |
| 2-Hydroxyethyl methacrylate (HEMA) | 〇 | 〇 | 〇 | 〇 | × | - | - | - |
| N-(2-Hydroxyethyl)acrylamide | 〇 | 〇 | 〇 | 〇 | 〇 | × | - | - |
| N,N-Dimethylacrylamide | 〇 | × | - | - | - | - | - | - |
| 4-Acryloylmorpholine | 〇 | × | - | - | - | - | - | - |
Measured at 25℃
*◎: Easy 〇: Good ×: No soluble -: No measurement
2. Excellent resistance to hydrolysis
- The obtained monomer has excellent resistance to hydrolysis in acidic to basic aqueous solutions.
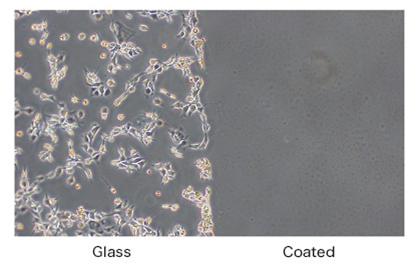
3. High biocompatibility
- It has high biocompatibility and resistant to cell adhesion and platelet adhesion.
*The inhibitory effect of coating on cell adhesion was examined by the FUJIFILM.
Example of use
- Capable of preparing highly hydrophilic functional coatings.
- Due to the high water-solubility of monomers and initiators, water may be used as a solvent for preparation to prepare coating solutions.
For specific examples, go to "Water-soluble cross-linker N-[tris(3-acrylamidepropoxymethyl)methyl]acrylamide".
Product List
- Open All
- Close All
For research use or further manufacturing use only. Not for use in diagnostic procedures.
Product content may differ from the actual image due to minor specification changes etc.
If the revision of product standards and packaging standards has been made, there is a case where the actual product specifications and images are different.
The prices are list prices in Japan.Please contact your local distributor for your retail price in your region.