PrimeSurface®


PrimeSurface®cultureware comprises ultra low attachment (ULA) dishes and plates that promote scaffold free, self assembly of spheroid formation. The plates are pre-coated with unique ultra hydrophilic polymer that enables spontaneous spheroid formation of uniform size and shape.
This cultureware is used for stem cell research, drug discovery & development, and tissue engineering & regenerative medicine.
Features
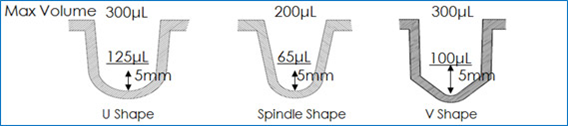
- The various well bottom shapes of 96 well plate ULA plates provide options for optimal spheroid growth for different cell types . Even though the U bottom plates are most widely used, the V bottom and M (Spindle) bottom wells would be a preferred choice for some cell types, especially when developing tight spheroids.
- Uniform single spheroid formation in each well
- Stable, non-cytotoxic and non-adhesive surface
- Easy handling, compatible with liquid robotic system
- Sterilized individual packaging
Spheroid formation (mouse ES cell)
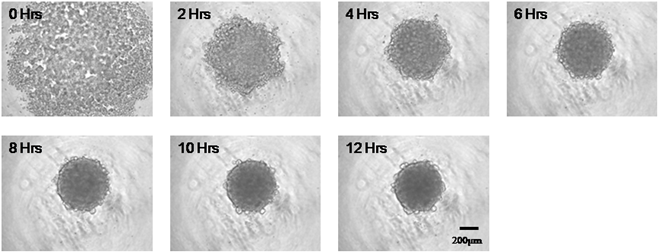
| Plate | PrimeSurface® MS-9096M |
|---|---|
| Cell | mouse ES cell 129SV |
| Quantity of cells | 1,500 cells/well |
| Culture Medium | DMEM + 4.5 mg/mL Glc. + 15%(v/v) inactivated FCS + 1%(v/v) NEAA + L-Glutamine(2mM) + β-Mercaptethanol(110µM/mL) + 1%(v/v) Pen.-Strep. |
| Culture Condition | 5% CO2, 37℃ |
PrimeSurface® 96 Slit-well Plate
A new design of ultra-low attachment 3D plate to facilitate easy handling of media exchange without disrupting spheroid formation.
Enhanced Stem Cell Culturing in Regenerative Medicine

Cell culturing involves frequent media replacement to provide nutrition to growing cells. In a standard 96 well ultra low cell attachment plate, media aspiration or dispensing has to be done extremely carefully to avoid disturbing the unattached spheroid, making this a time consuming operation. With the introduction of PrimeSurface® 96 Slit-well Plate, media exchange for 96 well plates can be efficiently handled with one step dispensing or aspiration for all 96 wells decreasing the pipetting time by over 80% while minimizing the risk of spheroid damage.
Product List
- Open All
- Close All
For research use or further manufacturing use only. Not for use in diagnostic procedures.
Product content may differ from the actual image due to minor specification changes etc.
If the revision of product standards and packaging standards has been made, there is a case where the actual product specifications and images are different.
The prices are list prices in Japan.Please contact your local distributor for your retail price in your region.



