LabAssay (TM) Ammonia
- for Cellbiology
- Manufacturer :
- FUJIFILM Wako Pure Chemical Corporation
- Storage Condition :
- Keep at 2-10 degrees C.
- GHS :
-
- Structural Formula
- Label
- Packing
- SDS
|
Comparison
|
Product Number
|
Package Size
|
Price
|
Inventory
|
|
|---|---|---|---|---|---|
|
|
|
700Tests
|
|
In stock in Japan |
Please check here for notes on products and prices.
Document
Kit component
For 700 tests
| Deproteinizing Reagent | 100 mL /1 bottle |
|---|---|
| Chromogen Reagent A | 50 mL /1 bottle |
| Chromogen Reagent B | 25 mL /1 bottle |
| Chromogen Reagent C | 50 mL /1 bottle |
| Standard Solution | 15 mL /1 bottle |
| Dilute Solution For Standard | 20 mL /1 bottle |
Product Overview
Ammonia is converted to urea in the urea cycle and excreted.
LabAssay™ Ammonia is a kit designed to measure ammonia based on a colorimetric method using the Berthelot reaction. This kit is used for the quantitative determination of ammonia in samples. It is a simultaneous multi-sample assay format using a microplate, but measurements can also be made using a test tube.
Kit Performance
| Analysis sample | Human Whole blood Mouse Whole blood Rat Whole blood Dog Serum/Plasma Cat Serum/Plasma Cell culture supernatants* |
|---|---|
| Calibration curve range | 100-400 μg/dL |
| Sample volume | 70 μL |
| Measurement duration | Approx. 70 min |
| Wavelength | 630 nm |
Example of Calibration Curve
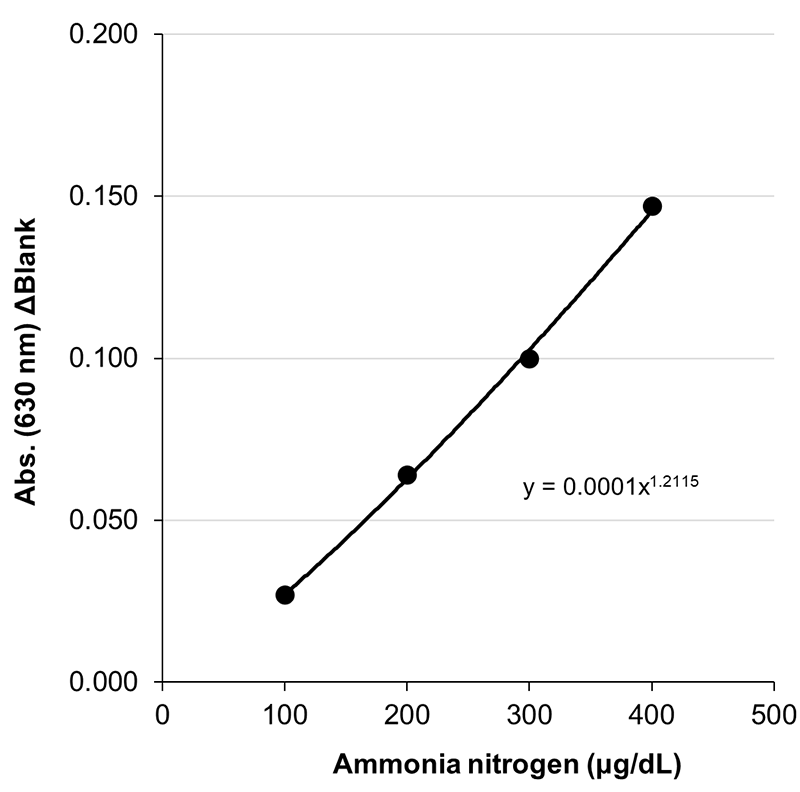
Assay principle
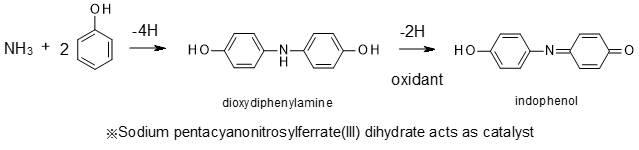
The Deproteinizing Reagent deactivates the enzymes and removes the components that inhibit color reaction in a sample. After adding Deproteinizing Reagent to a sample and centrifuging, add Phenol, Sodium Pentacyanonitrosylferrate(III) Dihydrate, and Sodium Hypochlorite to the supernatant. The indophenol blue pigment is obtained by the following schematic reaction. The amount of ammonia contained in the sample is determined by measuring the absorbance of the blue color.
Procedure
Assay in a microplate
Perform the assay in the wells according to the following table scheme.
| Test | Standard | Blank | |
|---|---|---|---|
| An experiment can be conducted in a microtube | |||
| Deproteinizing Reagent |
Deproteinizing reagent:Sample = 4:1(v/v) | Deproteinizing reagent:Standard solution = 4:1(v/v) | 70 μL |
| Sample | - | ||
| Mix well. Centrifuge 5000×g, 4℃, for 15 min. Subsequent operations should be performed in the wells. |
Mix well. Subsequent operations should be performed in the wells. |
||
| Supernatant 70 μL | Mixed solution 70 μL | - | |
| Chromogen Reagent A |
70 μL | 70 μL | 70 μL |
| Chromogen Reagent B |
35 μL | 35 μL | 35 μL |
| Chromogen Reagent C |
70 μL | 70 μL | 70 μL |
| After adding each reagent to a microplate, mix well and incubate at 37 ℃, for 20 min. Mix well, then let stand at room temperature* for 30 min. Mix well, then measure the absorbance of the test sample and standard solution at 630 nm with the blank as the control. |
|||
*Room temperature: 20℃ - 25℃
Data
Repeatability (within run precision)
Repeatability was conducted by measuring eight-fold assays of samples from mouse and rat with this product.
| n\ID | Mouse sample(BALB/c) | Rat sample(S.D. rat) |
|---|---|---|
| ID2 (μg/dL) | ID1 (μg/dL) | |
| 1 | 88.9 | 66.4 |
| 2 | 83.4 | 66.4 |
| 3 | 86.2 | 63.5 |
| 4 | 86.2 | 63.5 |
| 5 | 86.2 | 66.4 |
| 6 | 83.4 | 63.5 |
| 7 | 86.2 | 75.0 |
| 8 | 80.6 | 66.4 |
| mean | 85.1 | 66.4 |
| SD | 2.54 | 3.76 |
| CV(%) | 3.0 | 5.7 |
[Result]
The CV (%) of mouse sample was 3.0% and rat sample was 5.7%, indicating good repeatability.
Dilution Linearity Test
iPS cells were cultured in hPSC medium, and the ammonia concentrations in the culture supernatant were measured after 4-, 8-, 16-, and 32-fold dilution to evaluate linearity. The results for samples collected after 2, 4, and 5 days of culture are shown in the graph.
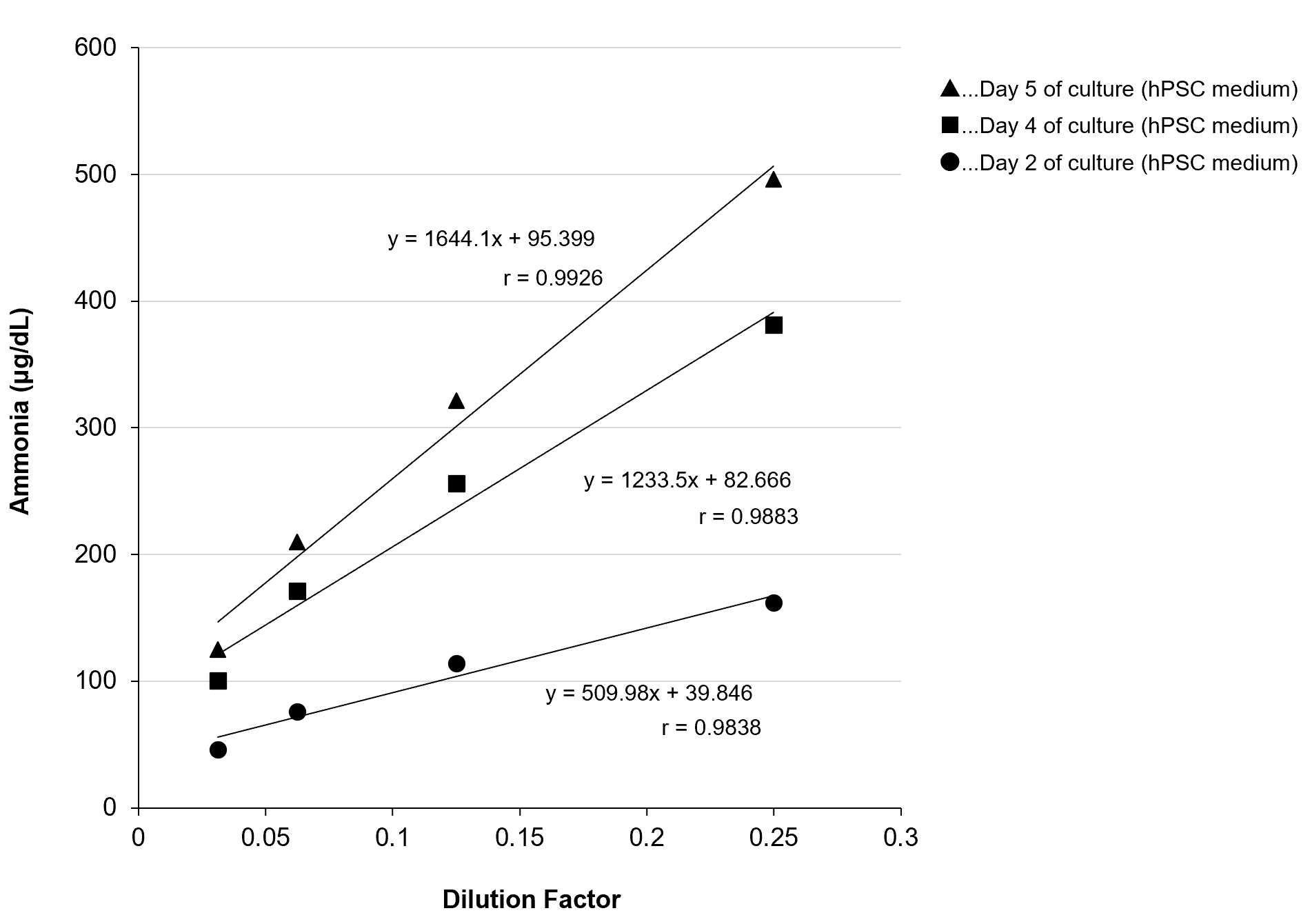
[Result]
All measurement results showed good linearity within the measurement range.
Correlation with Another Company’s Reagents for Benchtop Analyzers
Cell culture supernatant samples were measured using the plate assay method of this product and the tube assay method of reagents for benchtop analyzers from another company, and the correlation between the two sets of results was evaluated. For this product, the values obtained after 16-fold dilution were used based on the results of the dilution linearity study. The culture media used included hPSC (serum-free medium), D-MEM (10% FBS), E-MEM (10% FBS), and RPMI (10% FBS).
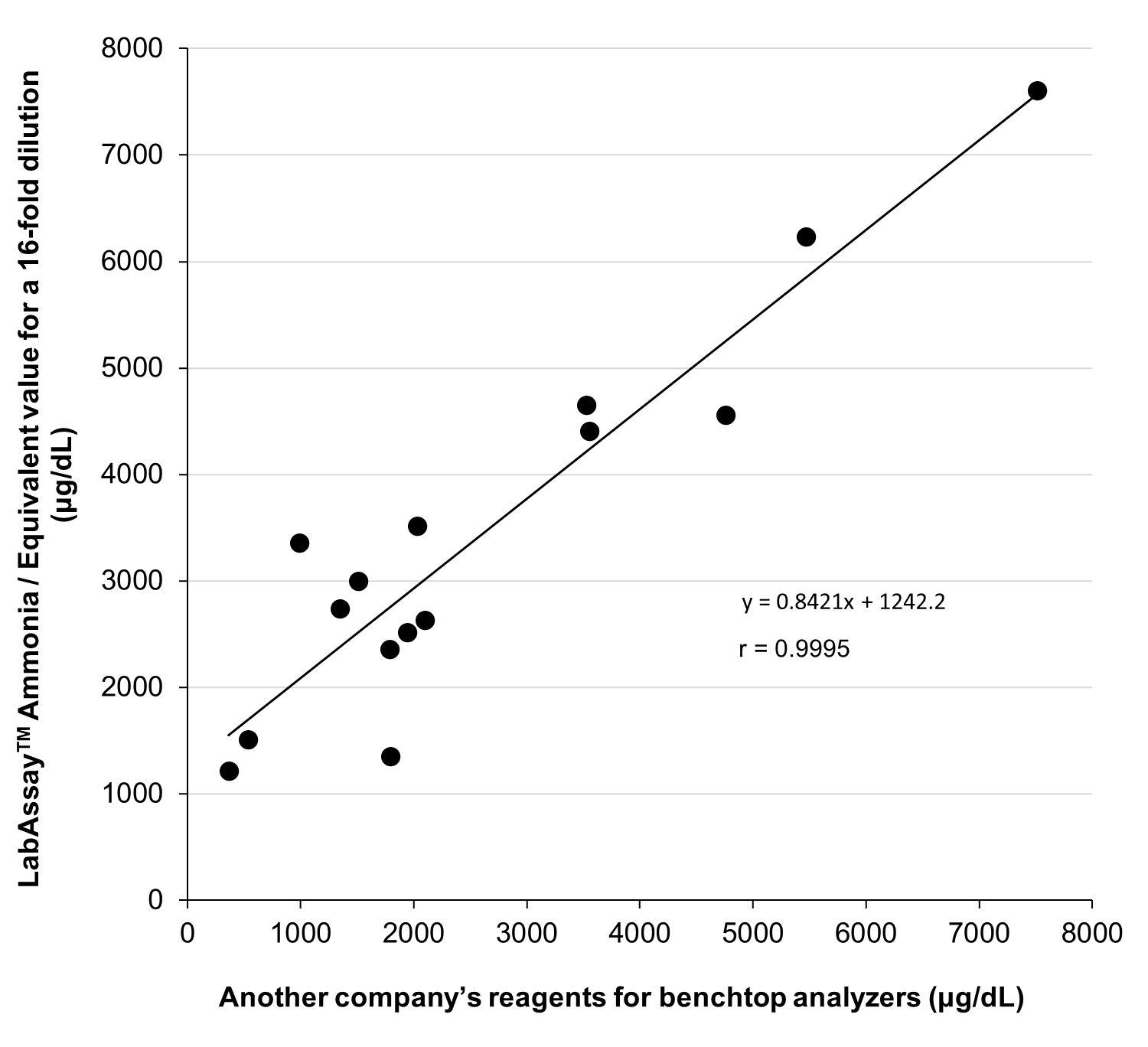
[Result]
A strong correlation was observed between the results obtained after 16-fold dilution with this product and those obtained using reagents for benchtop analyzers from another company.
Spike and Recovery Test
Standard solutions at three concentrations were added to samples from mouse and rat. Next, spike recovery tests were conducted, with each measurement performed in duplicate.
Mouse sample
| Spiked amount (μg/dL) |
Measurement value (μg/dL) |
Recovery volume (μg/dL) |
Recovery rate (%) |
|
|---|---|---|---|---|
| ID-a | - | 81.8 | - | - |
| 56.9 | 142 | 60.2 | 106 | |
| 114 | 205 | 123 | 108 | |
| 171 | 247 | 165 | 96.5 | |
| Average | 103 | |||
| ID-b | - | 89.2 | - | - |
| 56.9 | 139 | 49.8 | 87.5 | |
| 114 | 201 | 112 | 98.2 | |
| 171 | 230 | 141 | 82.5 | |
| Average | 89.4 | |||
| ID-c | - | 84.8 | - | |
| 56.9 | 145 | 60.2 | 106 | |
| 114 | 217 | 132 | 116 | |
| 171 | 255 | 170 | 99.4 | |
| Average | 107 | |||
Rat sample
| Spiked amount (μg/dL) |
Measurement value (μg/dL) |
Recovery volume (μg/dL) |
Recovery rate (%) |
|
|---|---|---|---|---|
| ID-a | - | 77.3 | - | - |
| 56.9 | 137 | 59.7 | 105 | |
| 114 | 188 | 111 | 97.4 | |
| 171 | 236 | 159 | 93.0 | |
| Average | 98.4 | |||
| ID-b | - | 77.3 | - | - |
| 56.9 | 135 | 57.7 | 101 | |
| 114 | 191 | 114 | 100 | |
| 171 | 242 | 165 | 96.5 | |
| Average | 99.3 | |||
| ID-c | - | 95.0 | - | |
| 56.9 | 147 | 62.0 | 109 | |
| 114 | 216 | 121 | 106 | |
| 171 | 265 | 170 | 99.4 | |
| Average | 104.8 | |||
[Result]
Good recovery rates were confirmed.
Measurement Using Actual Samples
Dog sample
| ID\n | Measurement value (μg/dL) |
mean | SD | CV (%) |
||
|---|---|---|---|---|---|---|
| 1 | 2 | |||||
| Serum | ID1 | 331 | 365 | 348 | 24.0 | 6.9 |
| ID2 | 296 | 323 | 310 | 19.1 | 6.2 | |
| ID3 | 409 | 465 | 437 | 39.6 | 9.1 | |
| ID4 | 367 | 365 | 366 | 1.41 | 0.4 | |
| ID5 | 453 | 513 | 483 | 42.4 | 8.8 | |
| ID6 | 349 | 366 | 358 | 12.0 | 3.4 | |
| ID7 | 451 | 504 | 478 | 37.5 | 7.8 | |
| ID8 | 291 | 323 | 307 | 22.6 | 7.4 | |
| Plasma (EDTA) |
ID1 | 232 | 210 | 221 | 15.6 | 7.0 |
| ID2 | 194 | 178 | 186 | 11.3 | 6.1 | |
| ID3 | 225 | 207 | 216 | 12.7 | 5.9 | |
| ID4 | 208 | 196 | 202 | 8.49 | 4.2 | |
| ID5 | 181 | 202 | 192 | 14.8 | 7.8 | |
| ID6 | 155 | 175 | 165 | 14.1 | 8.6 | |
| ID7 | 144 | 151 | 148 | 4.95 | 3.4 | |
| ID8 | 156 | 173 | 165 | 12.0 | 7.3 | |
Cat sample
| ID\n | Measurement value (μg/dL) |
mean | SD | CV (%) |
||
|---|---|---|---|---|---|---|
| 1 | 2 | |||||
| Serum | ID1 | 381 | 366 | 374 | 10.6 | 2.8 |
| ID2 | 370 | 378 | 374 | 5.66 | 1.5 | |
| ID3 | 381 | 342 | 362 | 27.6 | 7.6 | |
| ID4 | 357 | 329 | 343 | 19.8 | 5.8 | |
| ID5 | 249 | 245 | 247 | 2.83 | 1.1 | |
| ID6 | 315 | 299 | 307 | 11.3 | 3.7 | |
| ID7 | 403 | 388 | 396 | 10.6 | 2.7 | |
| ID8 | 198 | 193 | 196 | 3.54 | 1.8 | |
| Plasma (EDTA) |
ID1 | 191 | 204 | 198 | 9.19 | 4.7 |
| ID2 | 109 | 109 | 109 | 0.00 | 0.0 | |
| ID3 | 113 | 119 | 116 | 4.24 | 3.7 | |
| ID4 | 138 | 156 | 147 | 12.7 | 8.7 | |
| ID5 | 133 | 137 | 135 | 2.83 | 2.1 | |
| ID6 | 103 | 114 | 109 | 7.78 | 7.2 | |
| ID7 | 129 | 137 | 133 | 5.66 | 4.3 | |
| ID8 | 170 | 169 | 170 | 0.707 | 0.4 | |
FAQ
About sample
- Are there any precautions to take when handling samples?
- Blood ammonia levels increase over time if blood is left at room temperature after collection. Immediately transfer the blood into a test tube or blood collection tube prefilled with deproteinizing solution, mix thoroughly, and centrifuge without delay.
- What precautions should be taken when using blood additives (e.g., anticoagulants or amino acid preparations)?
- Do not use heparin or double oxalate as anticoagulants. Amino acid preparations may cause a negative bias and should be used with caution.
About kit usage
- What instruments, and equipment are required for the assay using this kit?
- The instruments and equipment required for the use of this kit are listed below.
- 96 well microplate (transparent type)
- Micropipette
- Plate mixer*
- Incubator maintaining at 37°C*
- Microplate reader with 630 nm wavelength filter
(* This is not necessary depending on the model of the microplate reader.)
(For Test Tube method)
- Test tube or microtube
- Pipette
- Incubator maintaining at 37°C
- Spectrophotometer with 630 nm wavelength filter
- What are the storage conditions and shelf-life guidelines for each reagent?
- Store at 2-10°C and do not freeze.
Overview / Applications
Property
Manufacturer Information
Alias
- LabAssay
633-51761 (Previous code)
LABNH3-M1 (Previous code)
295-78901 (Previous code)
For research use or further manufacturing use only. Not for use in diagnostic procedures.
Product content may differ from the actual image due to minor specification changes etc.
If the revision of product standards and packaging standards has been made, there is a case where the actual product specifications and images are different.
The prices are list prices in Japan.Please contact your local distributor for your retail price in your region.









